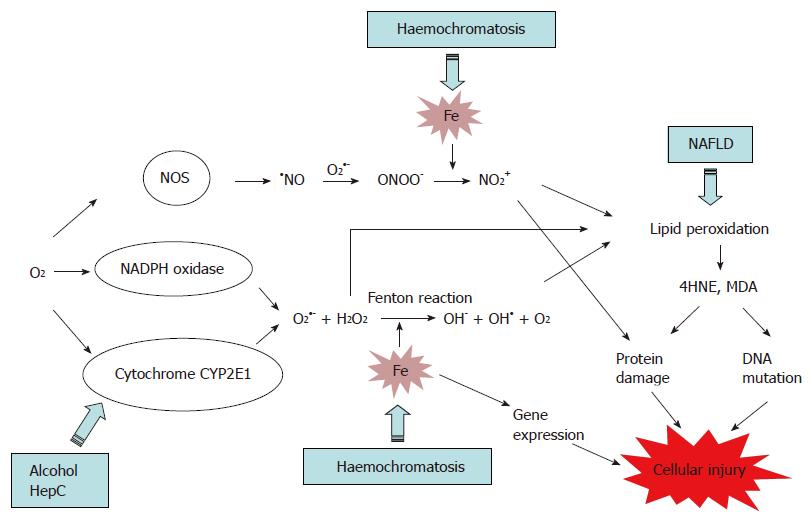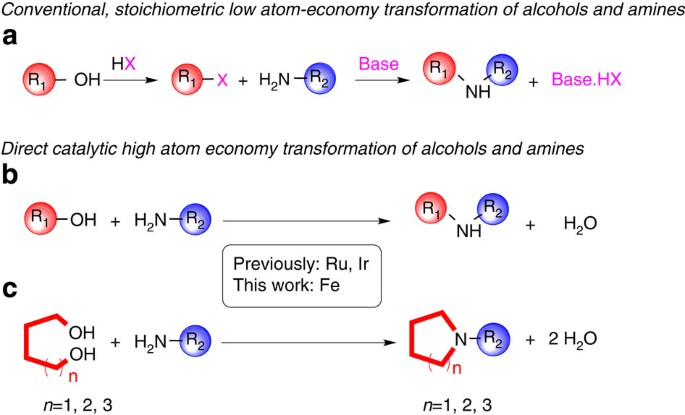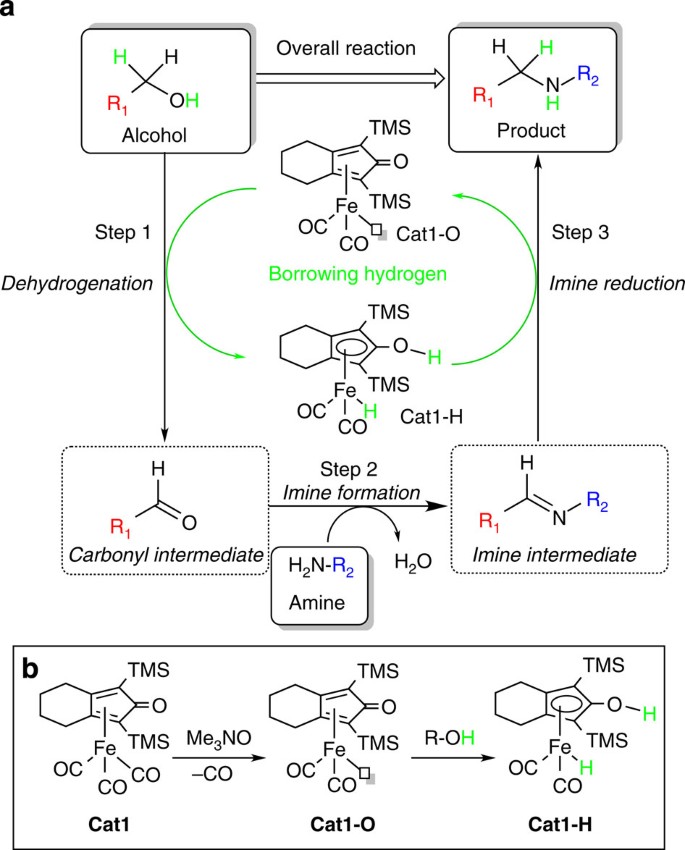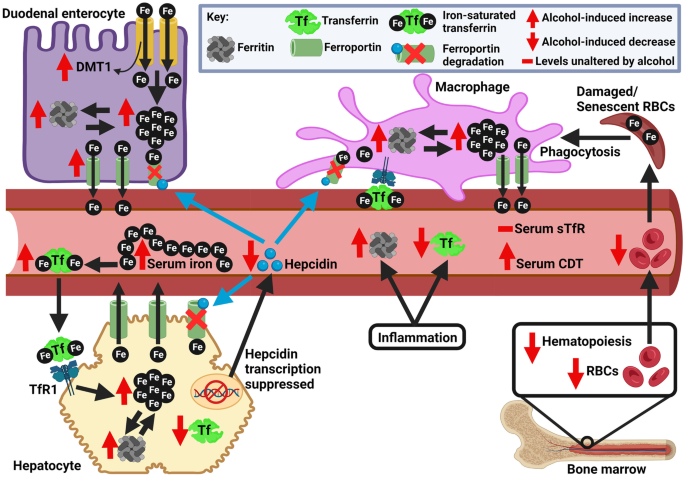
Iron and iron-related proteins in alcohol consumers: cellular and clinical aspects | Journal of Molecular Medicine

SOLVED: Does vitamin C speed up or slow down the reaction rate of iron contained inside produce with oxygen in the air? 2. Explain why a crushed tablet and a whole tablet

Reinvestigating FeBr3-Catalyzed Alcohol Oxidation with H2O2: Is a High-Valent Iron Species (HIS) or a Reactive Brominating Species (RBS) Responsible for Alcohol Oxidation? | Organic Letters

Investigation of iron oxide reduction by ethanol as a potential route to produce hydrogen - ScienceDirect
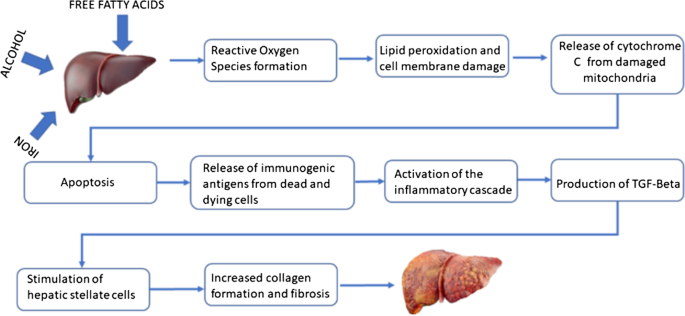
Hemochromatosis, alcoholism and unhealthy dietary fat: a case report | Journal of Medical Case Reports | Full Text

Molecules | Free Full-Text | A Simple Iron-Catalyst for Alkenylation of Ketones Using Primary Alcohols

Mechanism of the rearrangement of allyl alcohol with iron pentacarbonyl: evidence for a π-allyl-hydroirontricarbonyl complex - Chemical Communications (London) (RSC Publishing)
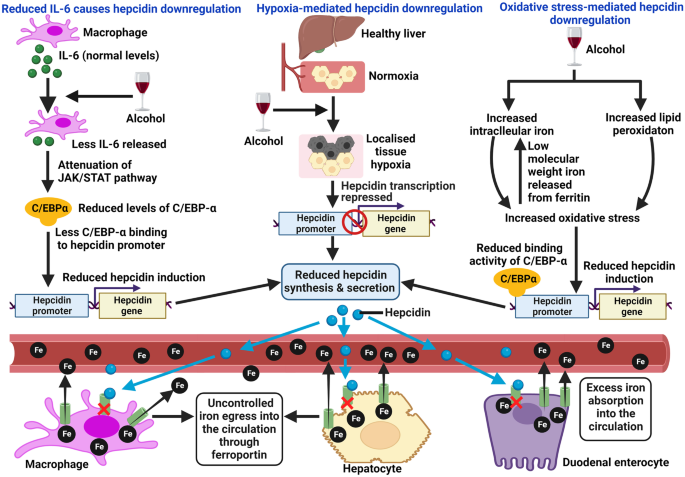
Iron and iron-related proteins in alcohol consumers: cellular and clinical aspects | Journal of Molecular Medicine
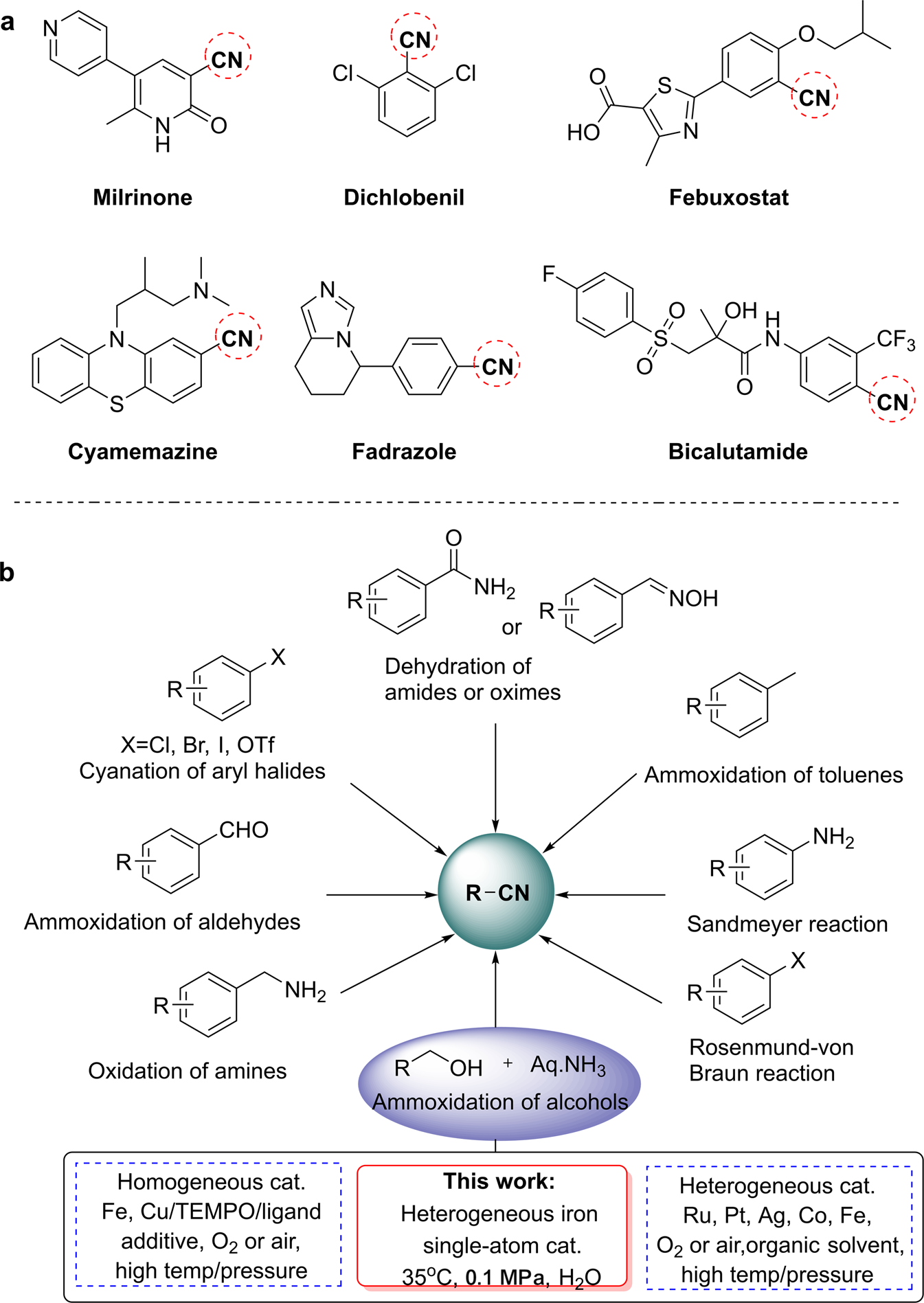
Efficient iron single-atom catalysts for selective ammoxidation of alcohols to nitriles | Nature Communications

Cyclopentadienone Iron Alcohol Complexes: Synthesis, Reactivity, and Implications for the Mechanism of Iron-Catalyzed Hydrogenation of Aldehydes | Journal of the American Chemical Society
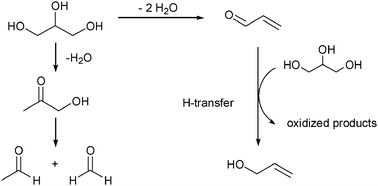
From glycerol to allyl alcohol: iron oxide catalyzed dehydration and consecutive hydrogen transfer - Chemical Communications (RSC Publishing)

Molecules | Free Full-Text | A Simple Iron-Catalyst for Alkenylation of Ketones Using Primary Alcohols

Alcohols as electrophiles: iron-catalyzed Ritter reaction and alcohol addition to alkynes - ScienceDirect

SOLVED: 59) Iron metal reacts with hydrochloric acid to produce iron (II) chloride and hydrogen gas. The balanced reaction that describes this process is: Fe + 2HCl â†' FeCl2 + H2. 60)